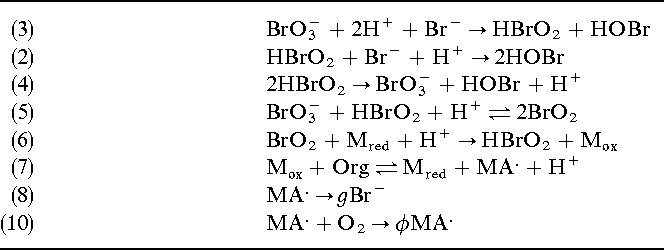Answer: The overall process involves applying redox (reduction-oxidation) reactions and balancing chemical equations using the ion-electron method (also known as the half-reaction method).
Explanation:
The sequence of equations describes a series of chemical reactions involving bromine species, hydrogen bromide, and oxidation states. The key concepts involved are:
- Redox reactions: Reactions where oxidation states change, involving electron transfer.
- Half-reactions: Separate oxidation and reduction processes, which are balanced separately.
- Ion-electron method: A systematic way to balance redox equations by balancing atoms and charge via electrons.
The steps involve identifying oxidation and reduction processes, writing half-reactions, balancing atoms and charges, and combining them to form the overall balanced equation.
Steps:
- Identify oxidation states and species involved:
- Bromine in \(\mathrm{BrO_3^-}\) (bromate ion) has an oxidation state of +5.
- Bromine in \(\mathrm{HBr}\) (hydrogen bromide) has an oxidation state of -1.
- Bromine in \(\mathrm{Br_2}\) (bromine molecule) has an oxidation state of 0.
- Bromine in \(\mathrm{HBrO_2}\) (hypobromous acid) has an oxidation state of +3.
- Write the oxidation and reduction half-reactions:
- Reduction: Bromate (\(\mathrm{BrO_3^-}\)) is reduced to bromine (\(\mathrm{Br_2}\)):
(This is a standard half-reaction for bromate reduction, but in the context of the sequence, it may be simplified or adapted.)
- Oxidation: Bromide (\(\mathrm{HBr}\)) is oxidized to bromine (\(\mathrm{Br_2}\)):
(or similar, depending on the specific reaction conditions.)
- Balance each half-reaction:
- For reduction:
- For oxidation:
- Combine the half-reactions:
- Equalize electrons:
- Multiply the reduction half-reaction by 2:
- Multiply the oxidation half-reaction by 5:
- Add:
- Cancel electrons and simplify as needed.
- Interpretation of the sequence:
- The equations (3)-(10) describe the stepwise transformations, including formation of intermediates like \(\mathrm{HBrO_2}\) and the involvement of \(\mathrm{H^+}\), \(\mathrm{Br_2}\), and \(\mathrm{BrO_3^-}\).
- The key method is balancing redox equations via half-reactions, considering oxidation states, and ensuring conservation of atoms and charge.
Summary:
The core mathematical concept is the balancing of redox reactions using the half-reaction method, which involves identifying oxidation states, writing separate oxidation and reduction half-reactions, balancing atoms and charge, and combining them to obtain the overall balanced chemical equation.