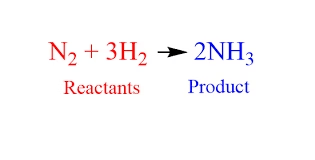
The balanced chemical equation is: $N_2 + 3H_2 \rightarrow 2NH_3$
Explanation
This is the Haber process for synthesizing ammonia. It shows that one molecule of nitrogen reacts with three molecules of hydrogen to produce two molecules of ammonia.
Steps:
- Recognize the reactants: nitrogen ($N_2$) and hydrogen ($H_2$).
- Recognize the product: ammonia ($NH_3$).
- Balance the equation:
- Nitrogen atoms: 2 on both sides (1 molecule of $N_2$ and 2 molecules of $NH_3$).
- Hydrogen atoms: 6 on both sides (3 molecules of $H_2$ and 6 atoms in 2 molecules of $NH_3$).
- The balanced equation is:
$$N_2 + 3H_2 \rightarrow 2NH_3$$
Therefore, the equation is already balanced as written.