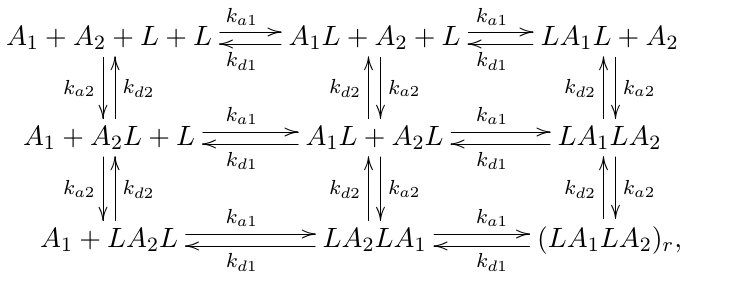Answer: The entire diagram represents a set of algebraic relations based on the properties of commutative and associative operations, involving reaction kinetics and rate constants in chemical reaction networks, specifically illustrating the detailed balance and equilibrium conditions in a reaction network.
Explanation:
This diagram encodes the relationships between different chemical species and their interactions via reaction rate constants ($k_{a1}$, $k_{a2}$, $k_{d1}$, $k_{d2}$) and the concentrations (or possibly activities) of species ($A_1$, $A_2$, $L$, etc.). It appears to be a reaction network with multiple pathways, illustrating forward and reverse reactions, and possibly the cycle conditions that relate these pathways, which are central to the theory of chemical kinetics and detailed balance.
The key concepts involved are:
- Reaction rate constants ($k_{a1}$, $k_{a2}$, $k_{d1}$, $k_{d2}$): parameters that determine the speed of reactions.
- Equilibrium constants: ratios of forward and reverse rate constants.
- Detailed balance: the condition where, at equilibrium, the rate of each forward reaction equals its reverse.
- Cycle conditions: relations that ensure the consistency of rate constants around a closed loop, often leading to the Wegscheider’s conditions.
- Associative and commutative properties: in the context of algebraic manipulations of reaction pathways.
The diagram shows multiple pathways connecting the same initial and final states, with ratios of rate constants, indicating the use of reaction network theory and graphical methods to analyze the system’s equilibrium or steady state.
Steps:
- Identify the species and reactions:
- The species involved are $A_1$, $A_2$, and $L$.
- The reactions involve the formation and dissociation of complexes like $A_1L$, $A_2L$, and their combinations, with rate constants $k_{a1}$, $k_{a2}$ (association), and $k_{d1}$, $k_{d2}$ (dissociation).
- Write the basic reaction rate equations:
For example, the forward and reverse reactions:
and similar for complex formations involving multiple species.
- Express the equilibrium relations:
At equilibrium, the ratio of concentrations (or activities) relates to the rate constants:
- Analyze the pathways:
The diagram shows multiple pathways connecting the same states, with ratios involving the rate constants. These pathways must satisfy cycle conditions for the system to be at equilibrium, which leads to relations such as:
ensuring detailed balance.
- Apply the principle of detailed balance:
The product of the ratios of forward to reverse rate constants around any closed loop must equal 1:
which is reflected in the diagram’s ratios.
- Relate to the algebraic expressions:
The expressions involving $A_1$, $A_2$, $L$, and their complexes, along with the ratios, encode these equilibrium conditions, ensuring the consistency of the network.
Summary:
The diagram is a graphical representation of the reaction network with multiple pathways, illustrating the kinetic relations and equilibrium conditions governed by reaction rate constants. The key theorems involved are the detailed balance theorem and the cycle condition in chemical kinetics, which ensure the consistency and thermodynamic feasibility of the network.
If you need further clarification or specific derivations of any part, please let me know!