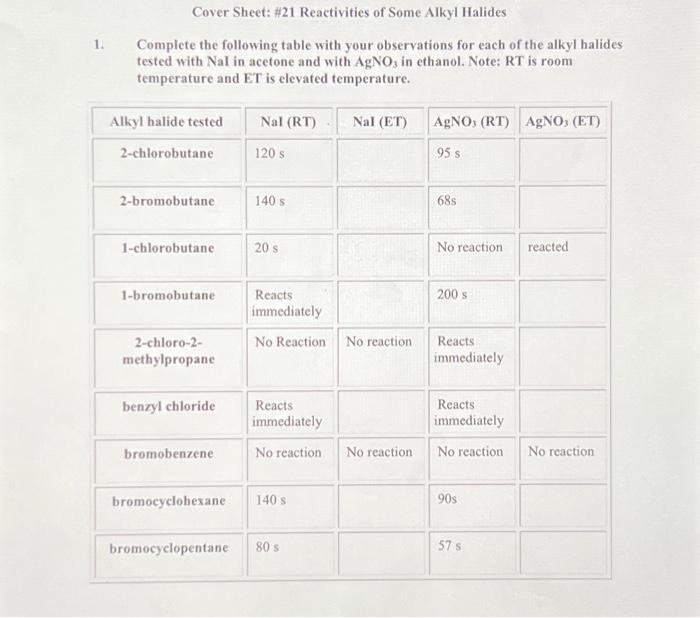
Answer: The subject is Chemistry, specifically the reactivity of alkyl halides in nucleophilic substitution reactions.
Explanation: This table involves testing the reactivity of various alkyl halides with sodium iodide (NaI) in acetone and silver nitrate (AgNO₃) in ethanol. The reactions are observed at room temperature (RT) and elevated temperature (ET). The reactivity is influenced by the structure of the alkyl halide and the type of halogen present.
Steps:
- Identify the Reaction Types:
- NaI in Acetone: This is a nucleophilic substitution reaction (S\(_N\)2 mechanism). Iodide ion (I\(^-\)) acts as a nucleophile, displacing the halide ion.
- AgNO₃ in Ethanol: This involves the formation of a precipitate of silver halide, indicating a substitution reaction (often S\(_N\)1 mechanism) where the halide ion leaves, forming a carbocation.
- Analyze the Reactivity:
- Primary Halides (e.g., 1-chlorobutane): React faster in S\(_N\)2 reactions due to less steric hindrance.
- Secondary Halides (e.g., 2-chlorobutane, 2-bromobutane): Show moderate reactivity.
- Tertiary Halides (e.g., 2-chloro-2-methylpropane): React quickly in S\(_N\)1 reactions due to stable carbocation formation.
- Aryl Halides (e.g., bromobenzene): Generally unreactive due to resonance stabilization and lack of carbocation formation.
- Complete the Table:
- Fill in the missing observations based on typical reactivity patterns and the given data.
- Consider temperature effects: Elevated temperatures generally increase reaction rates.
- Key Concepts:
- S\(_N\)2 Mechanism: Bimolecular nucleophilic substitution, favored by primary halides.
- S\(_N\)1 Mechanism: Unimolecular nucleophilic substitution, favored by tertiary halides.
- Steric Hindrance and Carbocation Stability: Major factors affecting reactivity.
By understanding these concepts, you can predict and explain the reactivity trends observed in the table.